Where does one start after a week like that?
The #FOAMed community is unique. Anyone who uses it regularly or contributes to it knows that. For most of us the knowledge we gain through social media and free sources drives our practice heavily. The format and environment helps us to stay motivated and always wanting to learn more. This passion that we have is what drives us to attend SMACC in the first place.
This year was my first SMACC experience. I couldn’t make it last year when it was in my own country. However, I think being in Ireland provided a unique aura. Ireland had a special meaning for FOAMed; it was born there, in a pub in downtown Dublin. In addition, one of the greatest FOAMed contributors and visionaries lived there. This of course was John Hinds. I won’t say much about him because anyone in this community must know who he was and I could never do him the justice he deserves. The conference this year had a certain feel too it. It seemed very appropriate to be there in honor of John. This was especially true at #SMACCforce.
There were many incredible pre-conference sessions offered. It was almost impossible to choose which to attend. The reputation of SMACCforce, the prehospital workshop, brought me there.
 A full day of education from some of the smartest, most motivated prehospital providers in the world. One of my favorite talks, an empassioned rant really, was from Per Bredmose (@VikingOne_). He spoke about the difficult situation of intervening when care is not appropriate or not aggressive enough. It really embodied the SMACC and FOAMed mantra of patient advocacy.
A full day of education from some of the smartest, most motivated prehospital providers in the world. One of my favorite talks, an empassioned rant really, was from Per Bredmose (@VikingOne_). He spoke about the difficult situation of intervening when care is not appropriate or not aggressive enough. It really embodied the SMACC and FOAMed mantra of patient advocacy.
“For fuck’s sake stand up for your patient!”
-Per Bredmose, The Viking
SMACCforce brings the importance of good prehospital critical care to the forefront. It is often overlooked, especially in the United States.
Monday night was also quite notable. During the trip I became quite involved with the EMS Wolfpack (@EMSWolfpack) and EMS Pocus (@emspocus, http://www.emspocus.com). The two groups collaborated to throw a pub night. We rented out an entire bar, The Trinity Inn, for a night of informal point-of-care ultrasound education, socialization, and alcohol consumption. We had no idea what to expect, nor did we know what was coming.

It started off with a few people learning the basics of POCUS (echo, RUSH, vascular access). But, before we knew it, the bar was full. It was impossible to even walk through it. In addition, Jim DuCanto (@jducanto) was there with his SALAD vomiting airway mannequin. This is probably the only documented instance of SALAD being performed in a bar; we are very proud of this. People were learning difficult airway techniques all throughout the night until the bar kicked us out.

However, the learning that was done was not the most remarkable point of the night. We had providers of all levels, from every continent, and from all specialities of critical care attend. People networked and collaborated with each other in a format that I believe is unique to SMACC. As was spoken about in SMACCforce, there was a true flattening of the heirarchy where paramedics were engaging physicians, and vice versa, in relevant and important conversation. The night was an overwhelming success and next year we will need a bigger pub.

Tuesday was the first day of the main conference. There were so many incredible talks that it would take me days to summarize them so I will focus on the ones that impacted me the most. The conference started with an incredible opening with a light show and the new smaccDUB soundtrack. This was followed by the John Hinds plenary, a series of speakers with topics designed to commemorate John.
Perhaps the most inspirational was Scott Weingart’s (@emcrit) talk where he encouraged us all the meditate. Scott has recently focused a lot on the psychological aspects of critical care and this is one way he believes that we can all improve our mental health and resuscitation abilities. There was also a video commemorating John’s life and a talk from his partner. I think there were tears throughout the auditorium.
“I will not leave you.”
-John Hinds to all of his patients
“Don’t be a resus wanker.”
-Also John
The rest of day one consisted of concurrent sessions. First I attended the Oxygen and Stress session where we learned from geniuses, including John Myburgh (@JAMyburgh) and self-proclaimed sex god Mervyn Singer that supplemental oxygen is probably a bad thing in most cases and patients do well without it.

The second half of the day was the How I Do It session. Providers spoke on specific practices they use regularly. Casey Parker (@broomedocs) discussed the importance of using evidence based values such as pretest probability when performing tests. In general many of the tests done in the emergency department are not helpful and difficult to interpret. Per Bredmose spoke on his use of inhaled nitric oxide in Norwegian helicopter critical care. Their use focuses primarily on bridging ARDS patients to ECMO but also in any cause of pulmonary hypertension such as pulmonary embolism. The day concluded with a panel discussing how medical knowledge and new data should be disseminated. There was argument about peer-review, journal bias, and the high cost to access new studies. The broad consensus was that we need a centralized, open access location to put all new data which has a publicly viewable peer-review process.
Day two started with a plenary called Slaying Sacred Cows. This was a series of four incredible talks which truly all deserve individual recognition. Resa Lewis (@ultrasoundREL) spoke inspirationally about women as leaders. She taught lessons that are designed to help both men and women succeed as leaders in medicine.
The talk that may have been the most impactful and lacrimation generating of the entire week was Ross Fisher’s (@ffolliet) Things That Scare Me, which he gave with no slides as the audience sat in intent silence.

Ross is a pediatric surgeon in the UK and he described some of the times during his practice that he has been most scared. This included operating on a 600 gram neonate with NEC. A patient this size has a blood volume of about 60 mL and they tend to bleed. He said that fear is the emotion that ends careers of excellent providers. The most important factor in dealing with fear is support from colleagues. In the right environment, fear can be overcome. This talk took such courage to give to a room of 2,000+. It is a must hear for everyone once it is released.
The next was from Reuben Strayer (@emupdates) who spoke about treatment of agitation. The most important part of the talk was this: we want droperidol back in the U.S.! It is a safe and effective drug for the significantly disruptive patient. If the patient is combative, or a threat to themselves or others, IM ketamine is the agent of choice.
The last talk of the morning plenary was from the amazing Liz Crowe (@LizCrowe2). She spoke about love in critical care and how it really impacts patient outcomes and provider health. As expected the talk was full of laughs and sexual enuendo. Remember your patients, patient families, and colleagues all need love.
The morning concurrent for me was All Heart. There were awesome talks on pediatric heart conditions, cardiac surgery patients, and the RV.

Haney Mallemat (@CriticalCareNow) intrigued the audience by proposing a modern approach to PEA arrest. Not all are the same and underlying causes must be identified and treated if ROSC is to occur. PEA is really an umbrella term, much like SVT. Ultrasound should be used to distiguish between pulselessness with rhythm and echo motion (PREM) and pulselessness with rhythm and echo standstill (PRES).
The afternoon of day two featured three different forums, of which I attended Bare Knuckle EM. There were four sessions which included an EM literature update. It turns out bacon can be used to treat epistaxis! Who knew? (http://www.ncbi.nlm.nih.gov/pubmed/22224315) Actual relevant topics were also discussed. The three following sessions were pro/con debates about important topics in EM. The topics were: EM is a failed paradigm, POCUS is a problem, and thrombolysis in submassive PE. Check these when they are released. The day concluded with SONIC BOOM, an entertaining and educational display of POCUS techniques.
We can’t get through day two without mentioning the incredible gala. The event was held at the Guinness Storehouse, the five floor venue where Guinness is made and sold in large quantities. Around 2,100 people attended and it was an awesome time.

The last day started off interestingly. The auditorium was about 1/4 full as a result of gala activities the night before. It gradually filled throughout the morning. The plenary talks were themed Beyond the Ivory Towers. The first two talks were from Kath Maitland (@KathMaitland) and Nikki Blackwell. They both discussed topics regarding their work in Africa providing care for critically ill patients with few resources. Kath focused on oxygen and its potential overuse.
Next, EM physcian Ashley Shreves discussed the immensely important topic of palliative care in the ED. This is done poorly pretty much everywhere. Incredible talk that will inspire you to do better.
“The treatment for death is probably not CPR in many cases!”
Ashley Liebig (@ashleyliebig) stole the show with her talk The Golden Fleece, The Golden Hour, and The Golden Rule.

In critical care, pre-hospital and in-hospital, providers carry a massive burden. Clinical responsibility paired with emotional trauma often leads to the downfall of incredible people. Ashley gave an emotional and honest talk with essentially no slides. I encourage everyone to listen to this when it is released.
Brining Sexy Back was the theme of my morning session. Weingart spoke about post-intubation sedation and analgesia. Remember: analgesia always and FIRST. The most notable talk though was from Alex Psirides (@psirides) on Why Your Hospital is Broken.

This talk was a reality check but true for most places. The focus of most hospital systems is often not truly the patient, it is the providers. Another must-hear.
My last concurrent of the conference was A is for Airway. Four great talks here. Reuben Strayer presented a somewhat novel approach to laryngoscopy designed to make providers slow down and not rush. Listen to the lecture and see what its all about.

The finale was a ton of fun. Two teams of four were pitted against eachother in contests testing knowledge, teamwork, and skills. All the participants earned half price admission to SMACC next year!
So in summary: epic. This post does not even scratch the surface. The talks were incredible. But what really makes SMACC special is the people you meet. And I can’t quanitify that in words. If you have not been to SMACC, go! Also, if you are not involved in #FOAMed and the twitter world I encourage you to start. Go to http://www.smacc.net.au and listen to the previous talks. Also, go to http://www.lifeinthefastlane.com, the original FOAMed site which has thousands of articles. Get on twitter, search #FOAMed, #smaccDUB, and get involved.

#dasSMACC 2017
Next year SMACC will be held in Berlin, Germany.
Venue: Tempodrom
June 26-29, 2017

![]() @amerelman
@amerelman




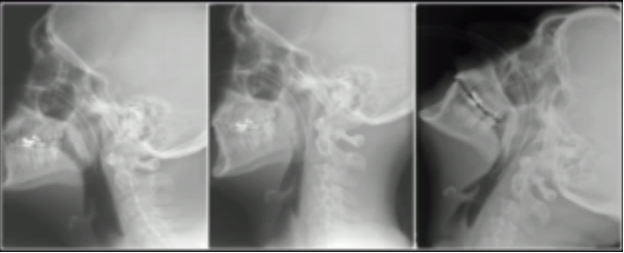
 So why is a bougie so useful? The objective answer is that it allows you to intubate patients with a more limited glottic view. As a general rule, it is best not to try to pass a styletted tube with a view that is worse than grade 2A. This is because anything worse does not allow you to visualize the tube entering the cords. Additionally, an ET tube is large and often obscures your view of the cords while trying to maneuver it to them.
So why is a bougie so useful? The objective answer is that it allows you to intubate patients with a more limited glottic view. As a general rule, it is best not to try to pass a styletted tube with a view that is worse than grade 2A. This is because anything worse does not allow you to visualize the tube entering the cords. Additionally, an ET tube is large and often obscures your view of the cords while trying to maneuver it to them.
 the larynx. This is obviously not optimal, as it is always best to try to visualize the cords. But if you can only see epiglottis after performing
the larynx. This is obviously not optimal, as it is always best to try to visualize the cords. But if you can only see epiglottis after performing 




 The optimal way to perform BVM ventilation is with two providers. A mask seal is held with both hands by one provider and the other squeezes the bag. When maintaining a mask seal with two hands a double C-E grip can be used. This allows both hands to be used for displacing the jaw forward and results in significantly improved mask seal. Another, often more effective, technique is placing the palms of both hands on the sides of the mask then using the index and other fingers to pull the jaw forward. This method may be preferred in difficult BVM situations. Maintaining a jaw thrust is essential to maximizing oxygenation. Otherwise the airway obstructs and prevents air passage.
The optimal way to perform BVM ventilation is with two providers. A mask seal is held with both hands by one provider and the other squeezes the bag. When maintaining a mask seal with two hands a double C-E grip can be used. This allows both hands to be used for displacing the jaw forward and results in significantly improved mask seal. Another, often more effective, technique is placing the palms of both hands on the sides of the mask then using the index and other fingers to pull the jaw forward. This method may be preferred in difficult BVM situations. Maintaining a jaw thrust is essential to maximizing oxygenation. Otherwise the airway obstructs and prevents air passage.


 effects. The first is that they become significantly harder to recruit and inflate. Once an alveoli is collapsed it requires much more pressure to reinflate it. Alveoli that are collapsed cannot perform gas exchange leading to worsened oxygenation and ventilation. Additionally, when atelectasis occurs alveoli become damaged, less effective, and may rupture. This is especially true in patients with lung disease. Because of this, a PEEP valve should be used on all BVMs and adjusted individually for each patient. The typical setting for healthy lungs is 5 CMH2O but this can be increased in certain situations. Patients with pulmonary edema or other causes of physiologic shunt often require more PEEP to oxygenate and recruit lung tissue. Do not be afraid to increase PEEP if the oxygen saturation is not improving and always use at least 5 CMH2O.
effects. The first is that they become significantly harder to recruit and inflate. Once an alveoli is collapsed it requires much more pressure to reinflate it. Alveoli that are collapsed cannot perform gas exchange leading to worsened oxygenation and ventilation. Additionally, when atelectasis occurs alveoli become damaged, less effective, and may rupture. This is especially true in patients with lung disease. Because of this, a PEEP valve should be used on all BVMs and adjusted individually for each patient. The typical setting for healthy lungs is 5 CMH2O but this can be increased in certain situations. Patients with pulmonary edema or other causes of physiologic shunt often require more PEEP to oxygenate and recruit lung tissue. Do not be afraid to increase PEEP if the oxygen saturation is not improving and always use at least 5 CMH2O.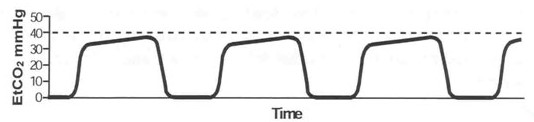
 this better than anyone, having examined 20 cadaveric airways per month for the past 16 years in addition to clinical research. He coined the term “coffin position” to describe the detriment of having your patient supine. Cannot Oxygenate Face Flat in Neutral position. There are a few reasons this is true and it is important to understand them. Not only will it help improve patient outcomes but it will make you better at managing airways.
this better than anyone, having examined 20 cadaveric airways per month for the past 16 years in addition to clinical research. He coined the term “coffin position” to describe the detriment of having your patient supine. Cannot Oxygenate Face Flat in Neutral position. There are a few reasons this is true and it is important to understand them. Not only will it help improve patient outcomes but it will make you better at managing airways.